Mold contamination in finished animal feed storage is a persistent technical challenge that leads to mycotoxin accumulation, nutrient degradation, and significant economic losses. This paper systematically examines the biological thresholds and environmental parameters that govern fungal growth in stored compound feed, and presents evidence-based control strategies spanning post-pelleting moisture management, organic acid preservative selection and dosing, storage facility design, and monitoring protocols.
Key quantitative data are presented throughout, including water activity (aw) thresholds for specific mold species, minimum inhibitory concentrations (MIC) of organic acids, cooling process specifications, and shelf life outcomes under varying humidity regimes. The goal is to provide feed technologists and quality managers with an actionable, data-supported framework for mold prevention across the full post-production storage chain.
1. Introduction
Mold growth in stored animal feed is one of the most economically significant quality failures in the feed industry. Once pelleted or mixed feed leaves the production line, it enters a storage environment that, without active management, will often favor fungal proliferation. Ambient humidity, residual heat in freshly produced pellets, improperly sealed packaging, poor warehouse ventilation, and inadequate use of chemical preservatives collectively create conditions that can reduce shelf life from months to days.
The consequences extend beyond visible spoilage. Mold species commonly found in feed—Aspergillus flavus, Penicillium spp., and Fusarium spp.—produce secondary metabolites known as mycotoxins, which are thermostable, invisible, and toxic to livestock even at sub-ppm concentrations. Aflatoxin B1 (AFB1) is a Group 1 human carcinogen; its EU maximum permitted level in complete poultry and swine feed is 0.01 mg/kg and 0.02 mg/kg respectively. Ochratoxin A (OTA), deoxynivalenol (DON), and zearalenone (ZEA) each carry species-specific regulatory limits that, once exceeded, require feed disposal.
This article provides a comprehensive, technically oriented guide to mold prevention in finished feed storage, organized around the three principal control levers: (1) reducing water availability through moisture management, (2) chemical inhibition via organic acid preservatives, and (3) environmental and packaging controls during storage.
2. Fungal Biology: Understanding What Enables Mold Growth
2.1 Water Activity as the Primary Limiting Factor
The single most important parameter governing mold growth in stored feed is not moisture content per se, but water activity (aw)—defined as the ratio of the partial vapor pressure of water in the feed to that of pure water at the same temperature (aw = p/p0). Water activity ranges from 0 (bone dry) to 1.0 (pure water) and directly represents the availability of water for biochemical and microbial processes.
Unlike moisture content (expressed as %), aw accounts for the fact that different feed matrices bind water differently. A feed with 13% MC may have an aw of 0.68 or 0.75 depending on its composition. For regulatory and safety purposes, aw is the operationally critical value.
Table 1. Minimum Water Activity (aw) Requirements for Growth of Key Mold Species in Animal Feed
| Mold Species | Minimum aw for Growth | Optimal aw Range | Primary Mycotoxin(s) | Typical Feed Matrix |
| Aspergillus flavus | 0.78 | 0.90–0.99 | Aflatoxins B1, B2, G1, G2 | Corn, groundnut meal, soybean meal |
| Aspergillus parasiticus | 0.78 | 0.90–0.95 | Aflatoxins B1, B2, G1, G2 | Corn, oilseed meals |
| Aspergillus ochraceus | 0.77 | 0.85–0.95 | Ochratoxin A (OTA) | Wheat, barley, mixed feeds |
| Penicillium verrucosum | 0.80 | 0.88–0.95 | Ochratoxin A (OTA) | Temperate cereal-based feeds |
| Fusarium graminearum | 0.90 | 0.95–0.99 | DON, ZEA, NIV | Wheat, corn (pre-harvest) |
| Fusarium verticillioides | 0.87 | 0.93–0.99 | Fumonisins B1, B2 | Corn-based compound feeds |
| Penicillium spp. (general) | 0.78–0.82 | 0.87–0.95 | Various (OTA, patulin) | Wide range; cold-tolerant |
| Xerophilic molds (e.g., Xeromyces bisporus) | 0.61 | 0.70–0.80 | Minimal toxin risk | Dry bakery by-products |
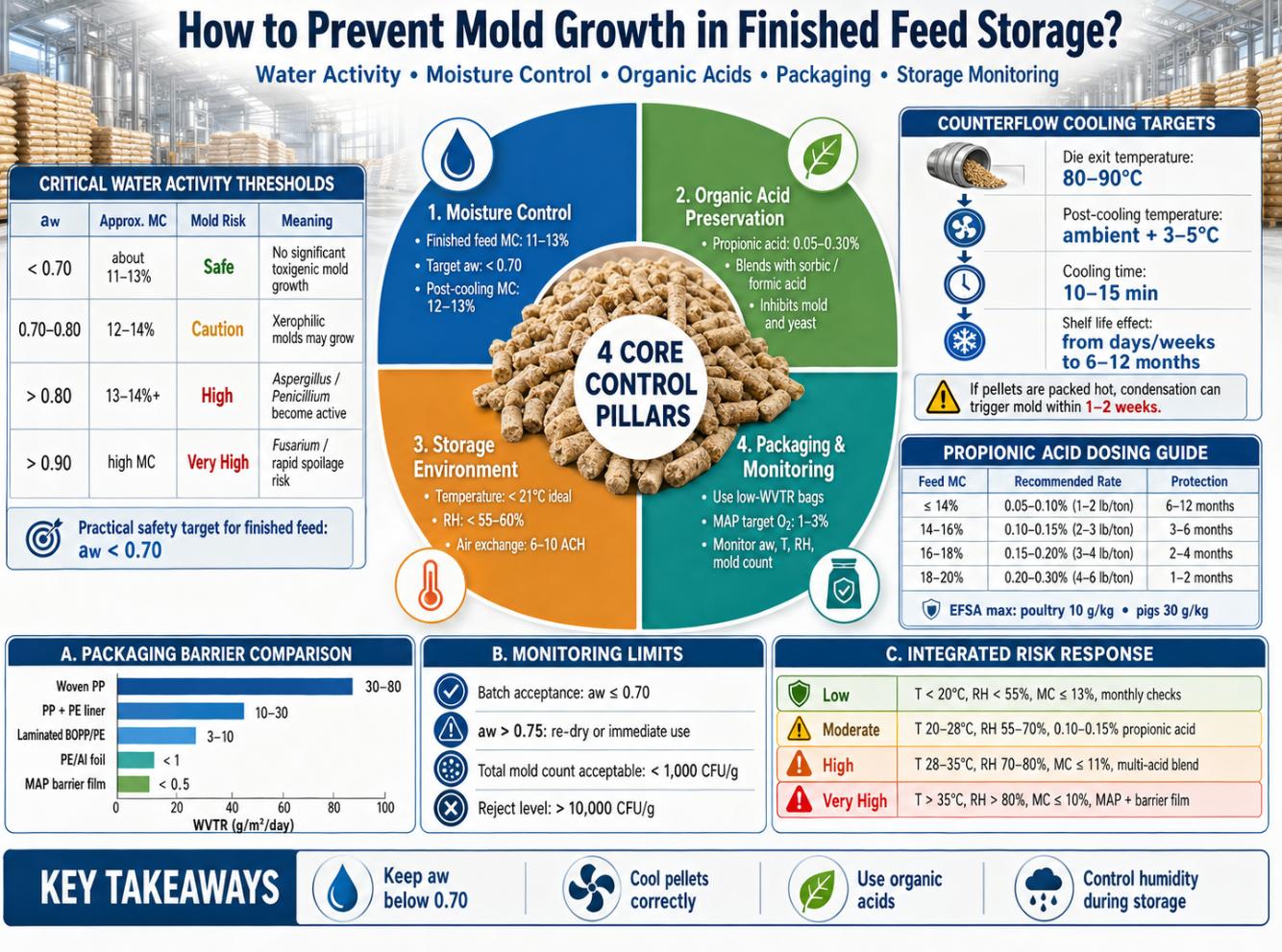
The critical practical threshold for most compound feeds is aw < 0.70. Below this level, no species of significant mycotoxigenic concern can grow. Between 0.70 and 0.80, xerophilic and xerotolerant species may germinate. Above 0.80, the most dangerous toxigenic molds—A. flavus and Fusarium spp.—become active. The stored grain moisture content threshold corresponding to aw ≈ 0.82 is approximately 13–14%, which is commonly cited as the maximum safe MC for stored grains and finished pellets.
2.2 Temperature and Its Interaction with Water Activity
Temperature and aw interact synergistically. Mold reproduction nearly stops at 2–5°C regardless of aw, and is significantly inhibited below 10°C. Most toxigenic storage molds are mesophilic, with optimal growth between 25–35°C. Keeping finished feed temperatures below 21°C substantially inhibits fungal growth; above 30°C, growth rates accelerate markedly even at borderline aw values.
Temperature also affects aw: for a given moisture content, aw increases with rising temperature. Feed stored at 35°C carries a materially higher mold risk than the same feed at 20°C with identical MC. In practice, feed warehouses in tropical climates (ambient 30–38°C, RH > 75%) represent the highest-risk storage environments and require the most aggressive combination of interventions.
Additionally, localized ‘hot spots’ within bagged or bulk-stored feed—caused by inadequate cooling before packaging, microbial respiration, or condensation—can sustain mold growth even when the average warehouse temperature is acceptable. This makes uniform cooling and temperature monitoring critical rather than optional.
2.3 Oxygen and CO2 Atmosphere
Most toxigenic feed molds are obligate aerobes requiring O2 concentrations above approximately 1–2% for germination and growth. Reducing headspace O2 to below 1% through nitrogen flushing or modified atmosphere packaging (MAP) effectively prevents mold establishment. CO2 at concentrations above 20% exerts additional fungistatic effects. This provides the mechanistic basis for MAP as a mold control strategy in packaged finished feed.
3. Strategy 1 — Moisture Control: The First Line of Defense
3.1 Target Moisture Content and Water Activity Specifications
The foundational mold prevention strategy is controlling finished feed MC before packaging or storage. Industry-wide targets for dry pelleted compound feed are MC 11–13% and aw < 0.70. The specific target should be set based on the expected storage duration and ambient conditions, using the lower end of the range for tropical environments or extended storage:
Table 2. Recommended Finished Feed MC and aw Targets by Storage Duration and Ambient Condition
| Storage Duration | Ambient RH (warehouse) | Target MC (%) | Target aw | Minimum Preservative Action |
| < 4 weeks | Any | ≤ 13% | ≤ 0.72 | Optional if aw < 0.70 |
| 1–3 months | < 65% RH | 11–12% | ≤ 0.68 | Recommended at 0.05–0.10% |
| 1–3 months | 65–80% RH | 10–11% | ≤ 0.65 | Required at 0.10–0.20% |
| 3–6 months | Any | ≤ 11% | ≤ 0.65 | Required at 0.15–0.25% |
| > 6 months | Controlled (< 60% RH) | ≤ 10% | ≤ 0.60 | Required + MAP packaging |
3.2 Counterflow Pellet Cooling: Process Parameters and Outcomes
Pellets exiting the die typically carry MC of 17–18% and temperatures of 80–90°C. At these conditions, mold and bacterial growth would commence within hours if feed were packaged directly. Counterflow cooling is the industry-standard method to reduce both parameters simultaneously.
In counterflow cooling, ambient air flows upward through a descending bed of hot pellets, creating a counter-current heat and mass transfer. The most important design parameters for moisture removal are the cooling-bed depth and the air-to-pellet mass flow rate ratio. Published research establishes that a flow rate ratio of 0.5 is insufficient for adequate cooling at a 0.30 m bed depth, while a bed depth of 0.15 m at a ratio of 1.1 achieves adequate results. A bed depth of 0.45 m maximizes total moisture loss.
Table 3. Counterflow Cooling: Input Conditions, Target Outputs, and Mold Risk Implications
| Parameter | Pre-Cooling (Die Exit) | Post-Cooling (Target) | If Target Not Met |
| Pellet temperature (°C) | 80–90°C | Ambient + 3–5°C | Condensation in packaging; mold within 1–2 weeks |
| Moisture content (%) | 17–18% | 12–13% | aw > 0.80; Aspergillus/Fusarium active |
| Cooling time (min) | — | 10–15 min (counterflow) | Shorter = insufficient drying |
| Pellet breakage | High (soft, hot) | ~30% reduction | Dust = elevated surface area for mold |
| Shelf life (sealed bag) | Days to weeks | 6–12 months | < 1 month without preservative |
A critical operational detail: the cooler itself must be cleaned regularly. Residual fines and feed dust accumulating inside the cooler can harbor mold colonies that contaminate successive production batches. Monitoring and maintaining cooler cleanliness is a prerequisite for the shelf life data above to hold.
4. Strategy 2 — Chemical Preservation: Organic Acid Mold Inhibitors
4.1 Mechanism of Action
Organic acids inhibit mold through the undissociated acid mechanism. At external pH values typical of feed (pH 5–7), a portion of the acid exists in the uncharged, undissociated form (HA), which can diffuse passively across the lipophilic fungal cell membrane. Once inside the cell, at the near-neutral intracellular pH (~7), HA dissociates to release H⁺ and the conjugate anion (A⁻). The resulting intracellular acidification disrupts enzymatic function and inhibits ATP synthesis, nutrient transport, and cell division. The anion also interferes with specific metabolic pathways (e.g., the citric acid cycle in Aspergillus). Critically, the efficacy of this mechanism is pH-dependent: the lower the feed pH (more acidic), the higher the proportion of undissociated acid and the greater the antifungal activity at a given total acid concentration.
4.2 Comparative Efficacy Data
A systematic plate assay study compared the minimum inhibitory concentrations (MIC) of eight organic acids against mold species commonly found in animal feed. The results established a clear efficacy ranking and identified practical application thresholds:
Table 4. Minimum Inhibitory Concentrations (MIC) of Organic Acids Against Feed Mold Species
| Organic Acid | MIC vs. Fusarium spp. (%) | MIC vs. Aspergillus spp. (%) | MIC vs. Penicillium spp. (%) | Relative Efficacy Rank |
| Propionic acid | 0.05–0.10 | 0.10–0.20 | 0.15–0.25 | High (2nd overall) |
| Valeric acid | 0.05–0.10 | 0.08–0.18 | 0.12–0.22 | Highest overall |
| Butyric acid | 0.05–0.12 | 0.10–0.20 | 0.15–0.25 | High (3rd overall) |
| Sorbic acid | 0.05–0.10 | 0.08–0.15 | 0.05–0.10 | High; best for Penicillium |
| Acetic acid | 0.30–0.80 | 0.50–1.50 | 0.80–2.00 | Moderate; requires high dose |
| Lactic acid | 0.50–1.20 | 1.00–2.50 | > 2.50 | Low; poor antifungal activity |
| Benzoic acid | 0.30–0.80 | 0.40–1.00 | > 2.00 | Moderate; limited vs. Penicillium |
| Formic acid | 0.05–0.15 | 0.20–0.50 | 0.40–1.00 | Strong vs. bacteria; moderate vs. molds |
The relative susceptibility of mold genera to organic acid inhibition follows the order: Fusarium spp. > Aspergillus spp. > Penicillium spp. This means that feeds in environments where Penicillium spp. are the dominant challenge (e.g., cool, temperate climates) require higher propionic acid inclusion rates or formulations enriched with sorbic acid. For yeast control specifically, acetic acid and sorbic acid are superior to propionic acid.
A multi-acid blend efficacy test comparing 75% buffered propionic acid against an 82% multi-acid blend (propionic + acetic + sorbic) at 2, 4, and 6 lb/ton found that at 4 lb/ton, the multi-acid blend completely suppressed wild yeast growth over a 12-hour incubation, while the single-acid product allowed rapid growth in the first 8 hours before suppression—underscoring the advantage of combined-acid formulations at moderate inclusion rates.
4.3 Application Rates and Regulatory Limits
Dosage selection must account for the feed MC at time of application, the target species, and applicable regulatory maxima. EFSA has established maximum safe levels based on long-term toxicological review:
Table 5. Propionic Acid Application Rates by Feed Moisture Content and Regulatory Maxima
| Feed / Grain MC (%) | Recommended Propionic Acid Rate | Equivalent Inclusion (g/kg) | Expected Protection Period | Notes |
| ≤ 14% | 0.05–0.10% (1–2 lb/ton) | 0.5–1.0 g/kg | 6–12 months | Standard dry feed; preventive dose |
| 14–16% | 0.10–0.15% (2–3 lb/ton) | 1.0–1.5 g/kg | 3–6 months | Adequate for temperate climates |
| 16–18% | 0.15–0.20% (3–4 lb/ton) | 1.5–2.0 g/kg | 2–4 months | High-humidity risk; blend preferred |
| 18–20% | 0.20–0.30% (4–6 lb/ton) | 2.0–3.0 g/kg | 1–2 months | Grain preservation; bridge to drying |
| 20–25% | 0.30–0.50% (6–10 lb/ton) | 3.0–5.0 g/kg | < 1 month | Wet harvest grain only |
| EFSA max: poultry | — | ≤ 10 g/kg complete feed | — | Regulatory ceiling (EFSA 2011) |
| EFSA max: pigs | — | ≤ 30 g/kg complete feed | — | Regulatory ceiling (EFSA 2011) |
Buffered forms (ammonium propionate, calcium propionate) reduce the corrosive and volatility issues associated with free propionic acid while maintaining comparable antifungal efficacy per unit of active acid. Calcium propionate is preferred for dry application in pelleted feed; ammonium propionate is common in liquid spray systems for grain treatment. For feeds with high fat content or where yeast is an additional concern, formulations combining propionic acid (0.10–0.15%) with sorbic acid (0.02–0.05%) and formic acid (0.05–0.10%) provide broader-spectrum protection.
5. Strategy 3 — Storage Environment and Packaging
5.1 Warehouse Design and Environmental Control
Even feed produced to exact moisture and preservative specifications can develop mold if the storage environment is poorly managed. The warehouse is the system within which all other interventions must function. Key environmental parameters and their targets:
Table 6. Recommended Storage Environment Parameters for Finished Feed and Risk Consequences of Non-Compliance
| Parameter | Target / Acceptable Range | High-Risk Condition | Mold Risk Consequence |
| Ambient temperature | < 21°C (< 70°F) | > 30°C | Growth rate doubles per ~10°C rise; Aspergillus spp. optimal |
| Relative humidity (RH) | < 55–60% | > 75% RH | aw re-equilibration through packaging; surface mold within days |
| Temperature uniformity | < 3°C variation across warehouse | Hot spots > 5°C above average | Localized condensation; mold foci that spread |
| Air exchange rate | 6–10 air changes/hour | < 3 changes/hour | Humidity and CO2 accumulation; accelerated spoilage |
| Ventilation timing (tropics) | Early morning / late evening only | Ventilation during peak RH | Moisture ingress into feed and packaging |
| Product stacking height | Per bag/sack spec; allow air gaps | Direct floor contact; wall contact | Condensation absorption; sealed moisture pockets |
| Datalogger monitoring interval | ≤ 15 minutes (T and RH) | Manual/daily checks only | Event detection delay; undetected excursions |
For facilities in tropical or subtropical climates where maintaining T < 21°C is impractical, the minimum effective target is T < 28°C combined with active dehumidification to maintain RH < 65%. At 28°C and 65% RH, well-preserved pellets (MC ≤ 11%, propionic acid ≥ 0.10%) can maintain acceptable mold counts for 8–12 weeks. Without active humidity control, safe storage duration shrinks to 4–6 weeks under these conditions.
5.2 Packaging: Moisture Barrier Selection
The packaging is the physical barrier between the feed and ambient humidity. Its performance is characterized by the Water Vapor Transmission Rate (WVTR), measured in g/m²/day at defined temperature and RH. Lower WVTR = better moisture barrier.
Table 7. Packaging Material Comparison: WVTR, Cost, and Application Suitability
| Packaging Type | WVTR (g/m²/day) | Relative Cost | Recommended Application |
| Woven PP (standard) | 30–80 | Low | Short-term storage (< 4 weeks), low-humidity environments |
| Woven PP + PE liner | 10–30 | Low–Medium | Up to 8 weeks; moderate-humidity environments |
| Laminated BOPP/PE | 3–10 | Medium | 2–4 months; standard commercial finished feed |
| PE/Al foil laminate | < 1 | High | Up to 12 months; premium/specialty/export feed |
| Multi-layer MAP (N₂ flushed) | < 0.5 (barrier film) | Very High | > 12 months; high-value or medicated feeds |
| Paper + PE + Al foil (tetra-type) | < 0.1 | Very High | Premix, vitamin concentrate, medicated feed |
For modified atmosphere packaging (MAP), the target headspace O2 concentration after gas flushing is 1–3%. Normal atmospheric O2 is 21%; at < 1%, aerobic mold growth is effectively eliminated. Nitrogen is the preferred purge gas (99%+ purity, food grade) as it is inert, non-toxic, dry, and inexpensive to generate on-site. CO2 additions at 20–40% of headspace volume provide additional fungistatic activity but must be used carefully in bags with rigid pellets, as CO2 is absorbed by feed over time, potentially causing package collapse.
5.3 Desiccants and Oxygen Absorbers
For high-value specialty feeds (premixes, vitamin-enriched feeds, medicated feeds, aquafeed with high lipid content), desiccant sachets and/or oxygen absorbers inserted within sealed packaging provide additional protection. Silica gel sachets (3–5 g per kg of product, depending on packaging WVTR and target shelf life) can maintain internal RH < 40% over 6–12 months. Iron-based oxygen absorbers (100–500 cc O2 capacity per sachet) reduce headspace O2 to < 0.1% within 24 hours of sealing, eliminating aerobic mold risk entirely for packaged products with adequate moisture barrier films.
6. Monitoring: Detecting Problems Before They Escalate
6.1 Water Activity Measurement
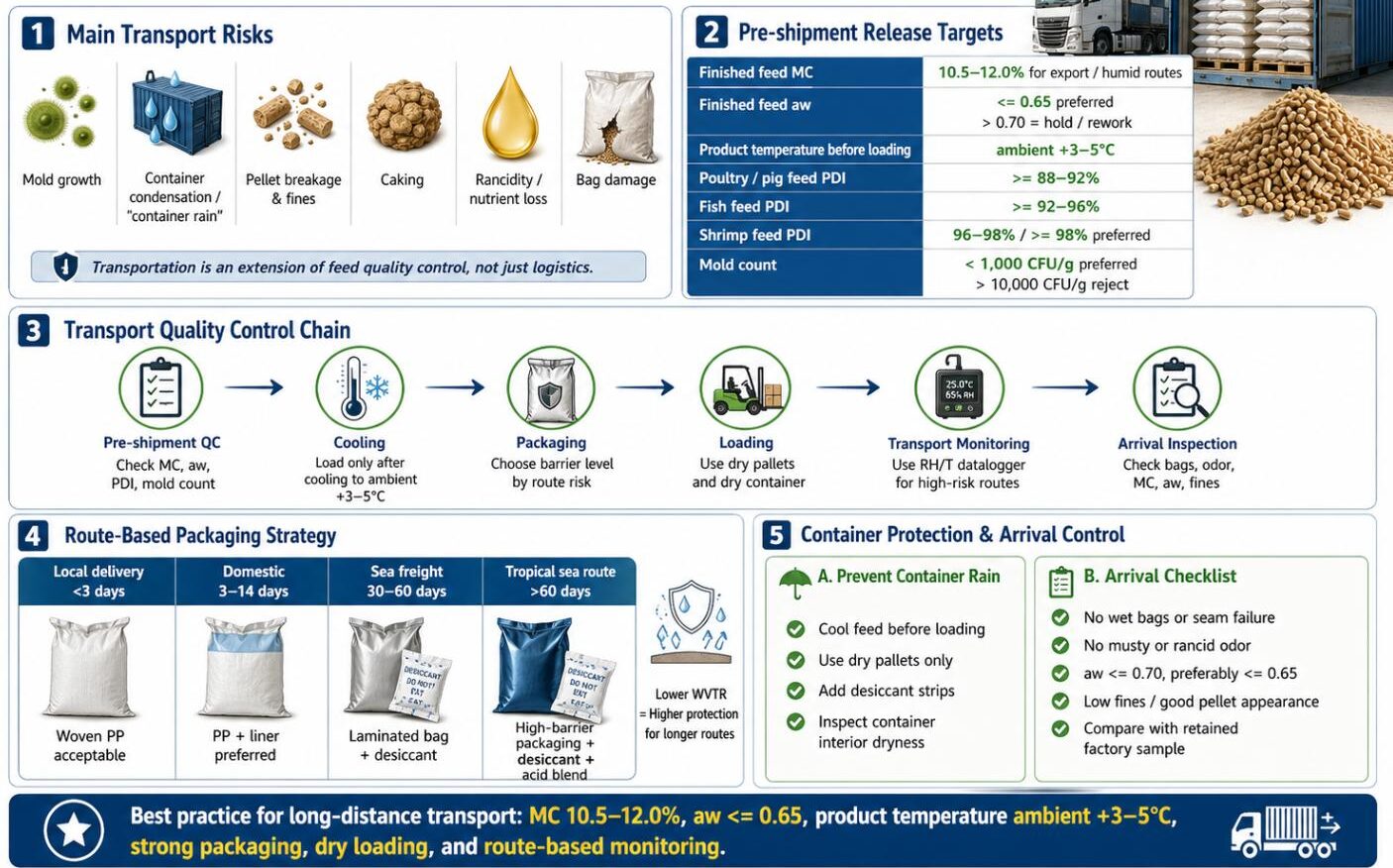
Regular aw monitoring of finished feed is the most direct indicator of mold risk. Portable aw meters (accuracy ±0.003 aw, equilibration time 5–15 min) are appropriate for routine QC at the mill and storage facility. Batch acceptance criteria should specify aw ≤ 0.70 for all finished feeds at point of packaging. Any batch exceeding aw 0.75 should be re-dried or diverted for immediate use.
In-line aw monitoring using capacitance-based sensors or microwave resonance sensors can be integrated into the post-cooler production flow, enabling real-time rejection of out-of-spec product before packaging. Microwave sensors offer the additional advantage of bulk measurement through the full pellet bed rather than surface-only readings, making them more representative for heterogeneous feeds.
6.2 Mold Count Testing
Routine fungal plate counts (total mold and yeast count, TMYC) on finished feed batches provide a microbiological indicator of process hygiene. Acceptance limits vary by species and application, but general guidelines for compound feed are:
Table 8. Microbiological Acceptance Criteria for Finished Compound Feed
| Indicator | Acceptable (CFU/g) | Alert Level (CFU/g) | Reject Level (CFU/g) | Action Required at Alert |
| Total mold count (TMYC) | < 1,000 | 1,000–10,000 | > 10,000 | Investigate source; increase preservative dose |
| Aspergillus spp. | < 100 | 100–500 | > 500 | Aflatoxin ELISA testing; review aw data |
| Fusarium spp. | < 100 | 100–1,000 | > 1,000 | DON/ZEA/fumonisin testing; check grain intake aw |
| Penicillium spp. | < 500 | 500–5,000 | > 5,000 | Review cooler hygiene; check storage RH |
| Total aerobic count | < 50,000 | 50,000–200,000 | > 200,000 | Hygiene audit; Salmonella testing |
It is important to note that mold count and mycotoxin level are decoupled: feeds with low mold counts may still contain significant mycotoxin concentrations if toxin-producing molds were previously active but have since died off. Conversely, feeds with elevated mold counts from non-toxigenic species may show acceptable toxin levels. Mycotoxin ELISA strip tests (on-site, semi-quantitative) and HPLC-MS/MS confirmation (laboratory, regulatory) should therefore be conducted independently from mold count results, especially for high-risk raw materials such as corn, groundnut meal, and cottonseed meal.
6.3 Inspection Protocol
Physical inspection of stored feed should follow a documented protocol covering the following checkpoints on a defined frequency (minimum weekly for hot/humid conditions, monthly for climate-controlled warehouses):
- Visual inspection for visible mold (white, green, black growth), caking, off-odors (musty, rancid, or ammonia-like), and discoloration on bag surfaces and in sampled bulk feed
- Temperature spot-checks using probe thermometers inserted into bulk stacks; temperature > 3°C above ambient in stored material indicates active microbial respiration
- Datalogger review for any T or RH excursions since last inspection; document maximum, minimum, and mean values
- Packaging integrity check for punctures, burst seams, or evidence of moisture condensation inside clear packaging
- FIFO (first-in, first-out) rotation audit to ensure oldest stock is dispatched first and no batch exceeds its calculated shelf life
7. Integrated Prevention Framework
Effective mold prevention in finished feed storage is not achievable through any single intervention. Table 9 summarizes the integrated decision framework that combines all three strategy pillars—moisture control, chemical preservation, and environmental/packaging management—calibrated to ambient storage risk level.
Table 9. Integrated Mold Prevention Framework by Storage Risk Level
| Risk Level | Ambient Conditions | Moisture Control | Preservative Strategy | Packaging | Monitoring Frequency |
| Low | T < 20°C, RH < 55% | MC ≤ 13%; aw ≤ 0.70 | Optional: 0.05% propionic acid | Standard woven PP | Monthly aw; quarterly mold count |
| Moderate | T 20–28°C, RH 55–70% | MC ≤ 12%; aw ≤ 0.68 | 0.10–0.15% propionic acid | Laminated PE/BOPP bag | Biweekly aw; monthly mold count |
| High | T 28–35°C, RH 70–80% | MC ≤ 11%; aw ≤ 0.65 | 0.15–0.25% multi-acid blend | Laminated + desiccant sachet | Weekly aw + mold count |
| Very High | T > 35°C, RH > 80% | MC ≤ 10%; aw ≤ 0.62 | 0.20–0.30% multi-acid blend | MAP (N₂ flush) + barrier film | Biweekly aw + mycotoxin ELISA |
A key principle underlying this framework is that interventions are additive in their efficacy but partially substitutable: higher preservative doses can partially compensate for marginally elevated aw, and superior packaging can extend shelf life even when warehousing conditions are suboptimal. However, no preservative dose or packaging system can compensate for severely elevated aw (> 0.80) or extremely poor storage conditions (T > 40°C, RH > 90%). At these extremes, the only effective intervention is re-drying or immediate use.
8. Conclusion
Mold prevention in finished feed storage is a multi-parameter engineering and management challenge. The biological evidence is clear: maintaining finished feed aw below 0.70 is the single most effective preventive measure. In practice, this requires consistent achievement of post-cooling MC of 11–13%, which in turn demands well-maintained counterflow coolers operating at the correct air-to-pellet flow rate ratio (≥ 1.1 for standard conditions) and bed depth (0.15–0.45 m depending on throughput).
Organic acid preservation, led by propionic acid and multi-acid blends, provides a critical second layer of defense—particularly in the inevitable scenarios where aw targets are marginally exceeded or where humidity ingress occurs during transport and distribution. The evidence-based dosage range for propionic acid in finished feed is 0.05–0.30% depending on MC and storage duration, with EFSA-established safety ceilings of 10 g/kg (poultry) and 30 g/kg (pigs) providing ample headroom for effective intervention doses.
Storage environment design—maintaining T < 21°C, RH < 60%, adequate ventilation, and appropriate FIFO rotation—closes the loop by ensuring that well-produced, well-preserved feed is not compromised during the storage and distribution phase. Packaging selection, from standard woven PP for short-term ambient storage to MAP and barrier laminates for extended or high-humidity applications, provides the physical boundary within which all other measures must function.
Together, these four pillars—moisture control, chemical preservation, storage environment management, and packaging—constitute a defensible, data-supported system for mold prevention across the full post-production storage chain.